|
Polarity of molecule results from non - symmetrical sharing of Valence Electron, creating a region of unequal charges in molecule. Therefore, dipoles of S - F bond do not cancel out each other and molecules turn out to be polar and contribute some dipole moment. So, is SF2 polar or nonpolar? Sf2 is polar in nature because sulfur and fluorine atoms in the molecule differ in their electronegativity and the molecule has a bent geometrical shape. 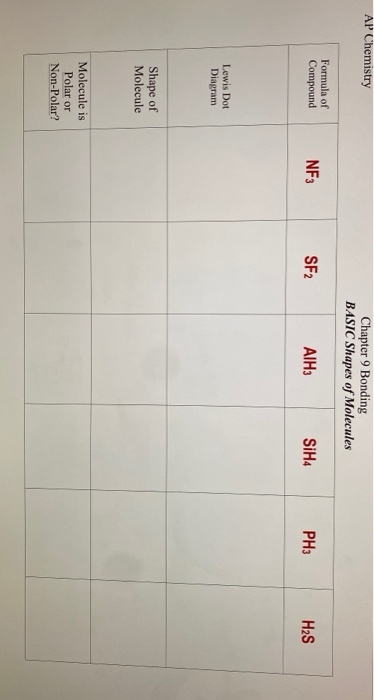
Compound can be polar or nonpolar depending upon sharing of charges on molecule, and this charge is spread because of sharing of electrons in the Valence Shell of the atom. However, biggest dilemma that students have about compound is its polarity. It comprises two atoms of fluorine attached to one atom of Sulfur. It is a compound that has a bond angle of 98 degrees between three atoms F - SF. Sulfur difluoride is a molecule denoted by chemical formula SF2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
